 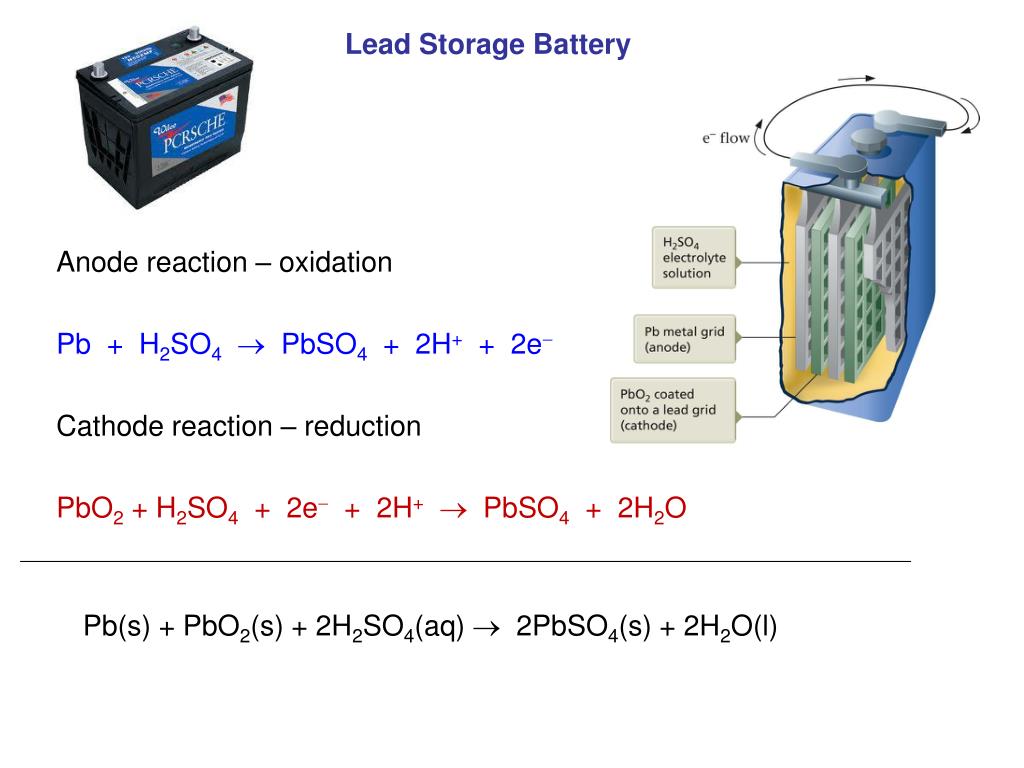 
In a nutshell, charging a battery converts electrical energy into chemical energy, while using (or discharging) a battery uses the stored chemical energy by converting it into electrical energy. In order to work again, the battery will need to be recharged, a process which is basically a reverse reaction of what we’ve just described. The battery will continue to work until it’s unable to supply enough power, a result of its electrolyte being depleted of sulphate ions. This surface area, while constantly giving off ions, will then continue to get smaller and smaller as more of its ions are used up. This decreases the area from which further electrochemical reactions can occur. As this happens, a series of sulphates are released and coat the lead plates of the anode and cathode. When a battery is either being discharged or used as a power source, the sulphuric acid in the electrolyte reorganises its ions so that it’s less concentrated, and more closely resembles H 2O. The electrochemical reaction facilitated by the battery acid allows a stable current to flow through the pairs of electrodes as electricity enters the anode and exits the cathode. This is usually a liquid solution of a solvent and dissolved salts An electrolyte, which allows ionic conduction to occur.A positive electrode or cathode, which r eceives electrons from the external circuit.A negative electrode, also known as the anode, which sends electrons to the external circuit.The common battery is usually composed of three essential parts: Put simply, battery acid facilitates the conversion of stored chemical energy into electrical energy. Battery acid is sulphuric acid that has been diluted with purified water What Does Battery Acid Do? 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
